A team from the College of Bioscience and Biotechnology at Yangzhou University published their findings in Angewandte Chemie International Edition (IF+12.3) where they report an engineered enzyme that can monooxygenate unprotected tryptophan into the corresponding 3a‐hydroxyhexahydropyrrolo[2,3‐b]indole‐2‐carboxylic acid (HPIC) in a single, scalable step with excellent turnover number and diastereoselectivity. Taking advantage of directed evolution, they analyzed the stepwise oxygen‐insertion mechanism of tryptophan 2,3‐dioxygenases, and transformed tryptophan 2,3‐dioxygenase from Xanthomonas campestris into a monooxygenase for oxidative cyclization of tryptophans. It was revealed that residue F51 is vital in determining the product ratio of HPIC to N′‐formylkynurenine. Their reactions and purification procedures use no organic solvents, resulting in an eco‐friendly method to prepare HPICs for further applications.
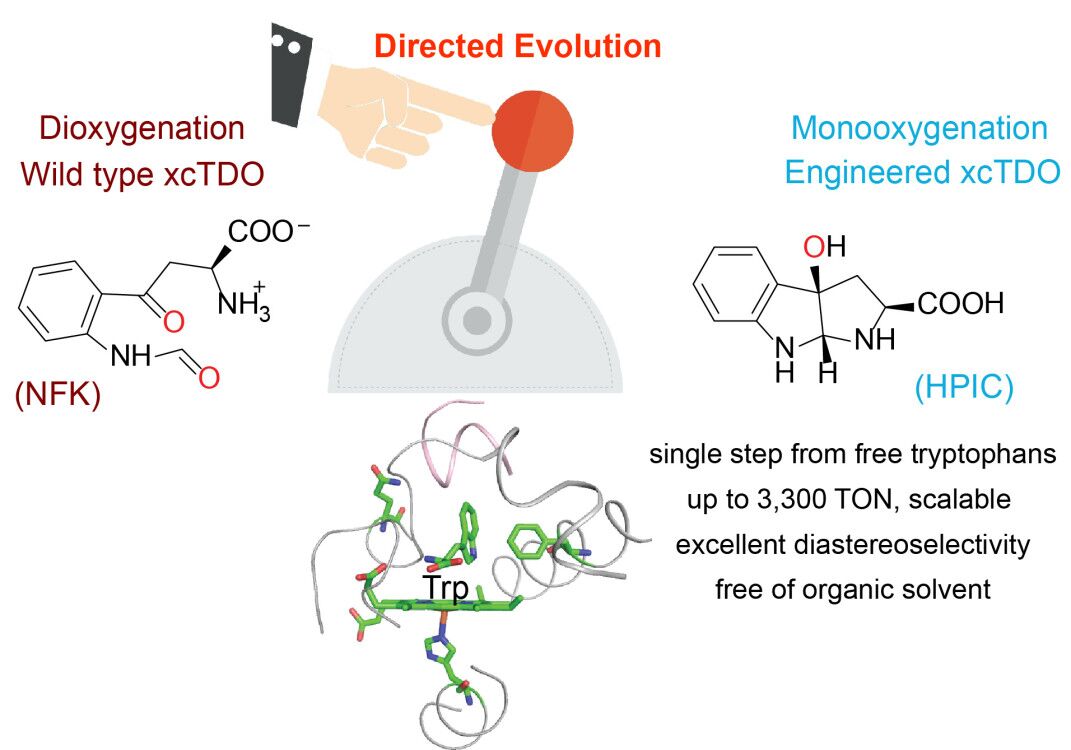
Wei Yanxin and Lu Chen, currently pursuing Master’ degree at the College of Bioscience and Biotechnology at Yangzhou University are the co-first authors of the paper. Dr. Wang Xiqing from Yangzhou University and Dr. Bai Wenju from Stanford University are the co-corresponding authors on the paper (Angew. Chem. Int. Ed. 2020, 59, 3043 –3047).

Read more: https://onlinelibrary.wiley.com/doi/10.1002/anie.201911825
![]() 苏公网安备 32100302010246号
苏公网安备 32100302010246号![]() 苏公网安备 32100302010246号
苏公网安备 32100302010246号